Hypoactive Sexual Desire Dysfunction Assessment and Management
Comprehensive, evidence-based insights and practical strategies for assessing and managing HSDD in clinical practice.
FREE CPD EDUCATION

Hypoactive Sexual Desire Dysfunction (HSDD) is a pervasive yet frequently unaddressed concern in clinical practice, significantly impacting patients' overall well-being and intimate relationships. Many clinicians, including
General Practitioners and Gynaecologists, recognise the need to enhance their understanding and management of this complex condition. Praxhub has curated this essential educational collection to provide a comprehensive and exceptionally valuable opportunity for you to upskill in HSDD, ensuring optimal support for affected patients.
With this education, you will gain a holistic, evidence-based understanding of HSDD, from its foundational definition and diagnostic criteria to advanced management strategies available in Australia and New Zealand. You'll gain practical tools for assessment and a framework for outlining diverse management options as well as an in-depth prescriber's guide to the TGA, Medsafe and globally registered treatment option, AndroFeme® 1.
Together, these activities will empower you to confidently address HSDD in your practice, leading to a profound and positive impact on your patients' quality of life.
About this education
Description:
HSDD is a common yet often under-recognised condition in primary care, most prevalent in post menopausal women. It is characterised by a persistent or recurrent lack of sexual interest, thoughts, or fantasies, and this absence leads to significant personal distress or difficulties in intimate relationships. Recognising and managing HSDD is crucial for primary care practitioners, as it can substantially impact patients' overall well-being and quality of life. Given the sensitive nature of sexual health, patients often find it easier to discuss these concerns initially with their trusted GP.
This module aims to equip primary healthcare practitioners with a comprehensive, evidence-based understanding of HSDD, including its definition, diagnostic criteria, potential causes, current treatment options available in Australia and New Zealand, and practical strategies for assessment and management in the primary care setting.
Learning outcomes:
- Define and explain Hypoactive Sexual Desire Dysfunction (HSDD).
- Identify the key factors contributing to HSDD in women, including biological, psychological, and interpersonal factors.
- Develop a framework for diagnosing HSDD, including the use of validated screening tools.
- Outline management options for HSDD.

ID: 1204
Steering Committee:
Hypoactive Sexual Desire Dysfunction: Assessment and Management in Clinical Practice has been endorsed by The Royal New Zealand College of General Practitioners (RNZCGP) and approved for up to 6 CME credits for Continuing Professional Development (CPD) purposes. Credits apply to active Fellows of the College only.
.png)
ID: 1275192
Integrated Activities:
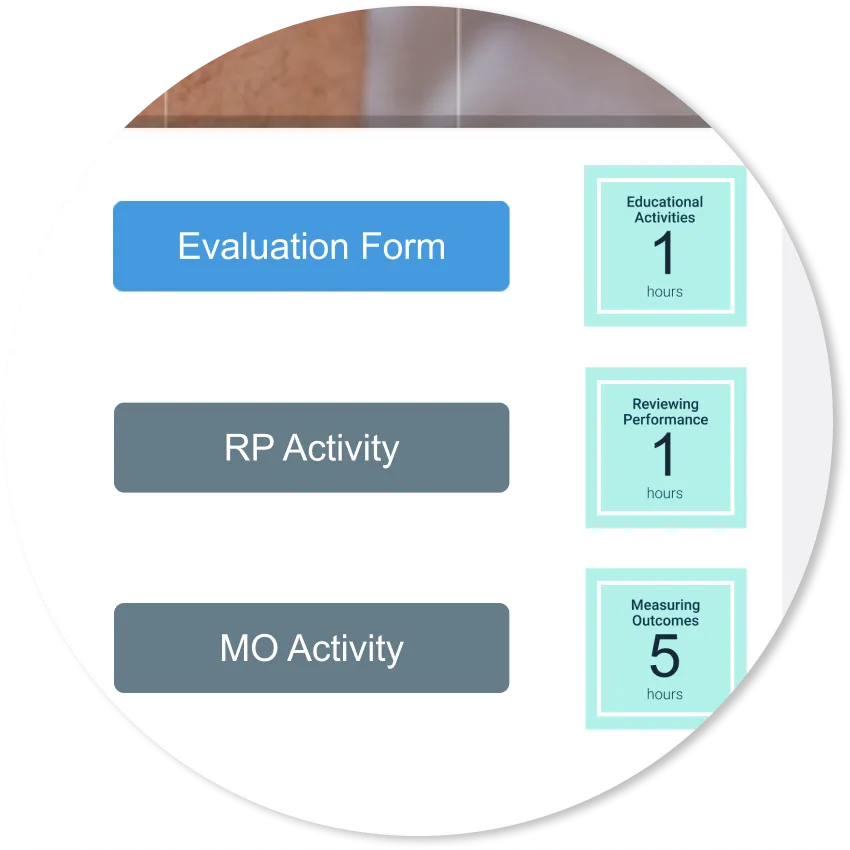
This activity offers up to 2.5 unique CPD hours:
- RP: 1 hr
- EA: 1.5 hrs
The series offers an additional 5 hr Measuring Outcomes activity
eLEARNING MODULE
Reviewing Performance (RP) Activity (1 hour)
This e-learning module is enhanced with a recommended but optional Reviewing Performance activity - approximate duration 1 hour.
Approx. duration 1 hour
Description:
This educational module offers a comprehensive overview of Hypoactive Sexual Desire Dysfunction (HSDD) and the role of AndroFeme® 1 in its management.
Specifically designed for postmenopausal women, AndroFeme® 1 stands as the sole TGA-approved/Medsafe registered product for HSDD treatment in Australia and New Zealand. Furthermore, it's the only globally approved solution for this condition. This module will delve into why AndroFeme® 1 is the only testosterone replacement scientifically proven to be both well tolerated and effective for women, having been specifically formulated for their unique needs.
This activity offers up to 2.5 unique CPD hours:
- RP: 1 hr
- EA: 1.5 hrs
The series offers an additional 5 hr Measuring Outcomes activity
eLEARNING MODULE
Praxhub would like to acknowledge Lawley for its support of this education. The sponsor has provided no input into the content of this activity.
To access hundreds of quality CPD training courses, simply join Praxhub's online medical community
© Praxhub 2026, all rights reserved.
- 'EA' stands for 'Educational Activities'
- 'MO' stands for 'Measuring Outcomes'
- 'RP' stands for 'Reviewing Performance'
Learn more about minimum EA, MO and RP requirements for General Practictioners.
Definitions
NZ Mandatories: AndroFeme®1 is a prescription medicine containing 1% w/v testosterone (10mg testosterone per 1 mL) indicated for the treatment of hypoactive sexual desire dysfunction (HSDD) in postmenopausal women. Therapeutic intervention with AndroFeme®1 should only be initiated in women following failure of appropriate education and correction of modifiable biopsychosocial factors (which may include neuroendocrine imbalance, physical ill health or disease, interpersonal difficulties, psychological distress or specific cultural or religious beliefs), according to the International Society for the Study of Women’s Sexual Health (ISSWSH) process of care. Testosterone supplementation in women must be monitored closely, especially at onset of treatment. It is recommended that women should ideally attend the same laboratory for baseline testosterone biochemistry prior to and during treatment. CONTRAINDICATIONS: AndroFeme®1 is contraindicated in patients with known sensitivity to testosterone, tree nuts (almond oil) or any of the excipients. It is contraindicated in females with known or suspected carcinoma of the breast, known or suspected androgen-dependent neoplasia, nephrotic syndrome, history of thromboembolism or hypercalcaemia. It is contraindicated in pregnancy and lactation and in women with normal reproductive function because of the potential for virilisation of a female fetus unless adequate contraceptive measures are being utilised. SPECIAL WARNINGS: Clinicians should refer to the data sheet for special warnings and precautions in relation to androgenic reactions, cardiac, hepatic or renal disease, use in athletes, inadvertent testosterone transfer, diabetes and potential thyroid effects. INTERACTIONS: No interaction studies have been performed with AndroFeme®1. There could be possible interactions with the following medicines: oral oestrogens, especially conjugated equine estrogen (CEE), tibolone and systemic glucocorticosteroids, anti-coagulants, ACTH or corticosteroids. Changes in insulin sensitivity, glucose tolerance, glycaemic control, blood glucose and glycosylated haemoglobin have been reported with non-oral androgen therapy. DOSAGE & ADMINISTRATION: Please refer to the data sheet for full information on dosing and administration. Before prescribing, please refer to the full Data Sheet available at www.medsafe.govt.nz. Unfunded Medicine – normal doctors’ fees and a prescription charge will apply. Alchemy Health NZ Ltd, Auckland. TAPS: NP24318

-v5.svg)

-v5.svg)